 Since its FDA approval in August of 2012 and subsequent launch in December 2012, nearly 1.5 million unique patients have filled nearly 7 million prescriptions for LINZESS, according to IMS Health. LINZESS® is the #1 prescribed brand for the treatment of irritable bowel syndrome with constipation (IBS-C) and chronic idiopathic constipation (CIC), based on IMS Health data. There are few available prescription treatment options for this condition. CIC is generally characterized by infrequent bowel movements (less than three times per week), but symptoms vary across this broad and heterogeneous patient population and may also include recurrent straining, lumpy or hard stools, and/or a sensation that the bowels are not fully empty. Phase III trials, spanning three doses and two indications.Ĭhronic idiopathic constipation (CIC) is a functional gastrointestinal disorder estimated to impact as many as 35 million adult Americans. LINZESS has met all primary endpoints in each of its five pivotal U.S. The most common adverse event was diarrhea the rates of diarrhea and of discontinuations due to diarrhea were numerically lower for the 72 mcg dose than the 145 mcg dose in this trial. As previously reported, this trial met its primary endpoint the 72mcg dose demonstrated statistically significant improvement in Complete Spontaneous Bowel Movements compared to placebo over 12 weeks. The approval of LINZESS 72 mcg is based on results from a Phase III clinical trial of 1,223 adults with CIC. 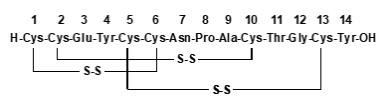
"LINZESS is the branded prescription market leader in the treatment of adult patients with IBS-C or CIC, and we believe the availability of a 72 mcg dose will enhance the product's utility to physicians in treating patients across the broad CIC patient population, which encompasses up to 35 million adult Americans," said Tom McCourt, Chief Commercial Officer at Ironwood.īill Meury, Chief Commercial Officer at Allergan, said, "This approval is testimony to the ongoing commitment of our organizations to continue to innovate with LINZESS and to enhance patient care and refine the treatment of gastrointestinal disorders such as CIC." Since the launch of LINZESS in December of 2012, nearly 1.5 million unique patients have filled nearly 7 million prescriptions, according to IMS Health data. LINZESS is now FDA-approved in three dosage strengths: 290 mcg for adult patients with irritable bowel syndrome with constipation (IBS-C), and 145 mcg and a new 72 mcg for the treatment of adults with CIC. The new dose is expected to be available in the first quarter of 2017.   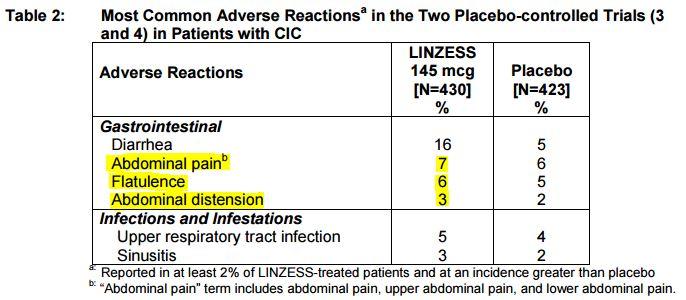
The newly approved dose will provide physicians with dosing flexibility based on individual presentation or tolerability, in treating the large and heterogenous population of adult CIC patients. Food and Drug Administration (FDA) has approved a 72 mcg dose of LINZESS ® (linaclotide) for the treatment of chronic idiopathic constipation (CIC) in adult patients. 26, 2017 /PRNewswire/ - Ironwood Pharmaceuticals, Inc. (NASDAQ: IRWD) and Allergan plc (NYSE: AGN) announced today that the U.S. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
